Interface and Surface Science for Safety Improvement
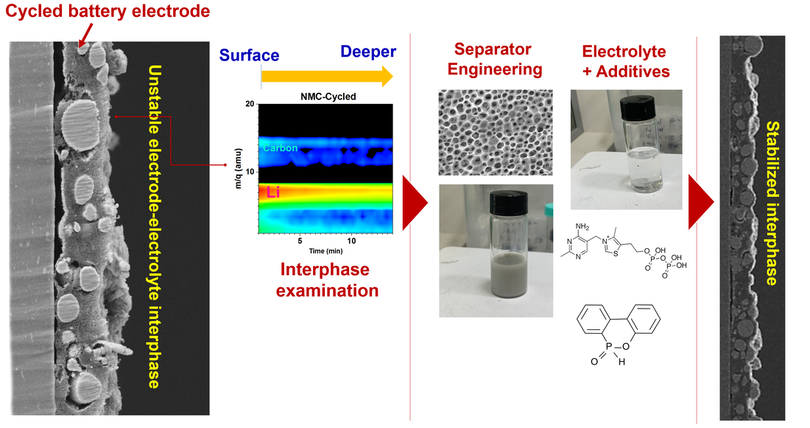
The interfacial issues within the electrochemical cells of lithium-ion batteries, lithium metal batteries, and other types of secondary batteries not only impact battery performance, but also pose significant safety concerns regarding potential fire hazards and even battery explosions. In particular, the unstable and thick development of the solid-electrolyte interphase (SEI) or cathode-electrolyte interphase (CEI) has adverse effects on battery cycling stability and performance. Furthermore, if metallic dendrites (such as Li-metal) grow actively at such interfacial conditions , they can cause internal short circuits, consequently increasing the risk of fire hazards. To address these issues, our research group focus to stabilize the electrode-electrolyte interphase employing engineered energy materials for lithium-ion batteries and similar energy storage applications.
Understanding Thermal Runaway: Crafting Effective Counter Strategies
What is a thermal runaway in energy storage applications ?
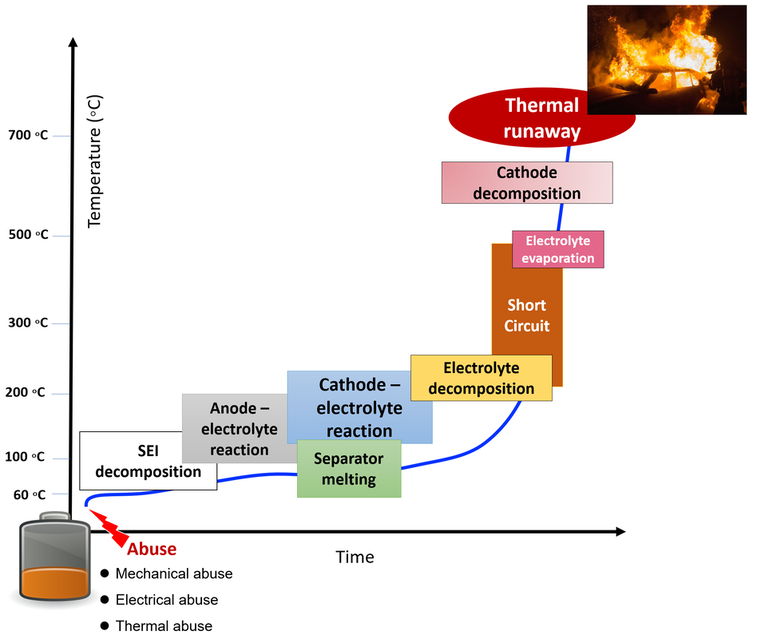
Thermal runaway in lithium-ion batteries refers to a self-reinforcing, uncontrollable increase in temperature within the battery cell. It typically begins with the overheating of one cell, which can lead to the release of heat and gas, causing neighboring cells to heat up as well. This process accelerates rapidly and can result in a violent and catastrophic failure, potentially leading to fire or explosion. Thermal runaway can be triggered by various factors such as overcharging, physical damage, manufacturing defects, or exposure to high temperatures.
Thermal runaway in lithium-ion batteries refers to a self-reinforcing, uncontrollable increase in temperature within the battery cell. It typically begins with the overheating of one cell, which can lead to the release of heat and gas, causing neighboring cells to heat up as well. This process accelerates rapidly and can result in a violent and catastrophic failure, potentially leading to fire or explosion. Thermal runaway can be triggered by various factors such as overcharging, physical damage, manufacturing defects, or exposure to high temperatures.
The thermal runaway chain reaction
In a lithium-ion battery, thermal runaway can set off a chain reaction due to the interconnected cell parts and the nature of the materials involved:
1. Initial Heat Generation: Thermal runaway often begins with a localized source of heat, which could be due to overcharging, internal short circuits, mechanical damage, or exposure to high temperatures (These are typically referred as abuse conditions).
2. Exothermic Reactions: As the temperature rises, the electrolyte and electrode materials within the battery undergo exothermic reactions. These reactions generate even more heat, further increasing the temperature of the cell.
3. Gas Generation: The elevated temperature can cause electrolyte decomposition and the breakdown of electrode materials, leading to the release of flammable gases such as hydrogen and carbon monoxide.
4. Pressure Build-Up: The generation of gases within the sealed battery enclosure leads to a rapid increase in internal pressure. This pressure build-up can result in the swelling or rupturing of the battery casing.
5. Propagation to Adjacent Cells: If the thermal runaway is not contained, the heat and pressure from the failing cell can spread to adjacent cells within the battery pack. This can trigger similar thermal runaway events in neighboring cells, leading to a domino effect.
6. Cascading Failure: As more cells undergo thermal runaway, the overall heat and pressure within the battery pack continue to increase, exacerbating the situation. This cascading failure can ultimately lead to a large-scale fire or explosion.
7. External Hazards: The release of flammable gases and the potential for fire or explosion pose significant external hazards, especially if the battery pack is located in a confined space or near flammable materials.
In a lithium-ion battery, thermal runaway can set off a chain reaction due to the interconnected cell parts and the nature of the materials involved:
1. Initial Heat Generation: Thermal runaway often begins with a localized source of heat, which could be due to overcharging, internal short circuits, mechanical damage, or exposure to high temperatures (These are typically referred as abuse conditions).
2. Exothermic Reactions: As the temperature rises, the electrolyte and electrode materials within the battery undergo exothermic reactions. These reactions generate even more heat, further increasing the temperature of the cell.
3. Gas Generation: The elevated temperature can cause electrolyte decomposition and the breakdown of electrode materials, leading to the release of flammable gases such as hydrogen and carbon monoxide.
4. Pressure Build-Up: The generation of gases within the sealed battery enclosure leads to a rapid increase in internal pressure. This pressure build-up can result in the swelling or rupturing of the battery casing.
5. Propagation to Adjacent Cells: If the thermal runaway is not contained, the heat and pressure from the failing cell can spread to adjacent cells within the battery pack. This can trigger similar thermal runaway events in neighboring cells, leading to a domino effect.
6. Cascading Failure: As more cells undergo thermal runaway, the overall heat and pressure within the battery pack continue to increase, exacerbating the situation. This cascading failure can ultimately lead to a large-scale fire or explosion.
7. External Hazards: The release of flammable gases and the potential for fire or explosion pose significant external hazards, especially if the battery pack is located in a confined space or near flammable materials.
Counter the safety concerns
Our group focus on developing materials with enhanced thermal stability, improved conductivity, and intrinsic safety features. Here are the major research topics:
[ Electrolyte Optimization ] Research efforts focus on developing electrolytes with higher thermal stability to withstand elevated temperatures without undergoing decomposition. This includes the exploration of electrolyte additives and formulations that improve thermal conductivity and suppress the formation of reactive species.
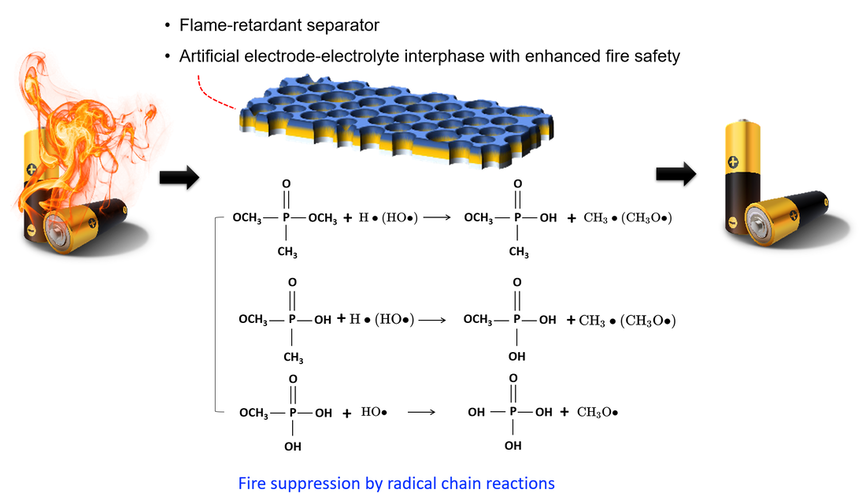
[ Separator Materials Engineering ] Enhance the thermal stability and mechanical strength of separator materials to prevent internal short circuits and thermal runaway propagation between battery electrodes. Nanostructured and ceramic-coated separators are being investigated to improve thermal resistance and inhibit dendrite formation.
[ Development of Electrode Materials with Enhanced Safety ] Develop cathode and anode materials with enhanced thermal stability and reduced reactivity with electrolytes. Incorporate thermally conductive additives into electrode formulations to facilitate heat dissipation within the battery cell.
[ Research on Artificial Cathode-Electrolyte/Solid-Electrolyte Interphase (CEI/SEI) with Flame Retardant Capability ]
The SEI (Solid Electrolyte Interphase) and CEI (Composite Electrode Interphase) are crucial components in lithium-ion batteries that form on the surface of the electrodes during the initial cycles of charging and discharging. These interphases serve as a protective layer that facilitates ion transport while preventing further reactions between the electrolyte and electrode materials. Hence, our attention is also directed towards engineering artificial SEI or CEI layers to enhance battery safety. This involves researching additives or functional groups that could be integrated into the artificial interphase, aiming to confer fire-retardant characteristics, minimize side reactions at the interphase, mitigate lithium dendrite formation, and enhance the mechanical and chemical attributes of SEI/CEI layers.
[ Electrolyte Optimization ] Research efforts focus on developing electrolytes with higher thermal stability to withstand elevated temperatures without undergoing decomposition. This includes the exploration of electrolyte additives and formulations that improve thermal conductivity and suppress the formation of reactive species.
[ Separator Materials Engineering ] Enhance the thermal stability and mechanical strength of separator materials to prevent internal short circuits and thermal runaway propagation between battery electrodes. Nanostructured and ceramic-coated separators are being investigated to improve thermal resistance and inhibit dendrite formation.
[ Development of Electrode Materials with Enhanced Safety ] Develop cathode and anode materials with enhanced thermal stability and reduced reactivity with electrolytes. Incorporate thermally conductive additives into electrode formulations to facilitate heat dissipation within the battery cell.
[ Research on Artificial Cathode-Electrolyte/Solid-Electrolyte Interphase (CEI/SEI) with Flame Retardant Capability ]
The SEI (Solid Electrolyte Interphase) and CEI (Composite Electrode Interphase) are crucial components in lithium-ion batteries that form on the surface of the electrodes during the initial cycles of charging and discharging. These interphases serve as a protective layer that facilitates ion transport while preventing further reactions between the electrolyte and electrode materials. Hence, our attention is also directed towards engineering artificial SEI or CEI layers to enhance battery safety. This involves researching additives or functional groups that could be integrated into the artificial interphase, aiming to confer fire-retardant characteristics, minimize side reactions at the interphase, mitigate lithium dendrite formation, and enhance the mechanical and chemical attributes of SEI/CEI layers.